|
4/17/2024 0 Comments Dalton's atomic theory states 
Priestley called the gas he discovered dephlogisticated air, but this name would not stick. For example, a colorless, odorless gas could combine with mercury, a silver metal, to form mercury calx, a red mineral. Priestley's discovery revealed that substances could combine together or break apart to form new substances with different properties. After many long days and nights in the laboratory, Priestley said of the strange gas, "What surprised me more than I can well express was that a candle burned in this air with a remarkably vigorous flame." Not only did flames burn strongly in this gas, but a mouse placed in a sealed container of this gas lived for a longer period of time than a mouse placed in a sealed container of ordinary air. Priestley carefully collected this gas in glass jars and studied it. Priestley had observed that it does not just turn into mercury it actually breaks down into two substances when it is heated, liquid mercury and a strange gas. Mercury calx, a red solid stone, had been known and coveted for thousands of years because when it is heated, it appears to turn into mercury, a silver liquid metal. Shortly after Bernoulli proposed his theory, the Englishman Joseph Priestley began to experiment with red mercury calx in 1773. Many scientists were busy studying the natural world at this time. Bernoulli reasoned that if these particles were not in constant motion, they would settle to the ground like dust particles therefore, he pictured air and other gases as loose collections of tiny billiard-ball-like particles that are continuously moving around and bouncing off one another. The particles could not be felt because unlike a solid stone wall that does not move, the tiny particles move aside when a human hand or body moves through them. 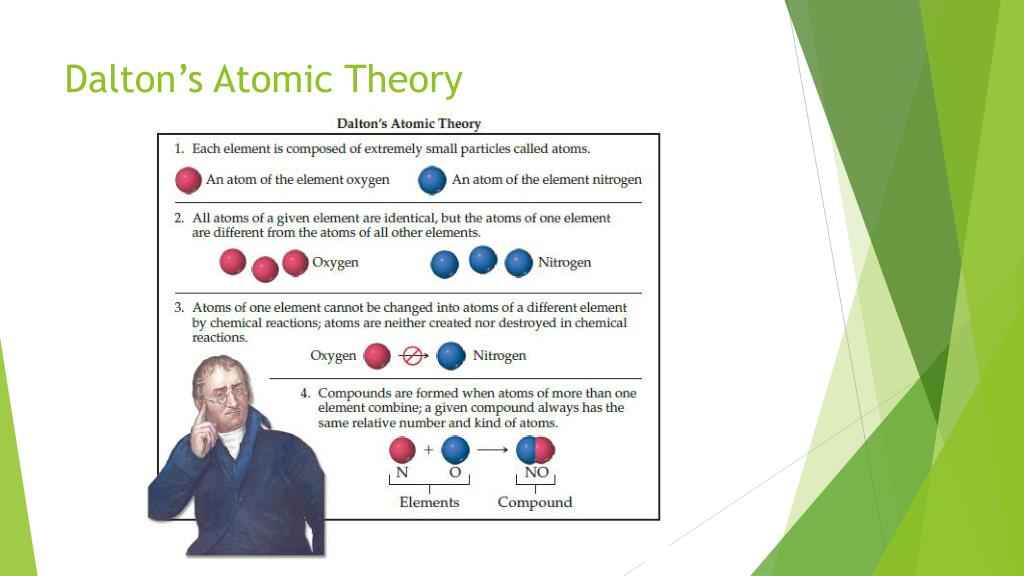
He developed a theory that air and other gases consist of tiny particles that are too small to be seen, and are loosely packed in an empty volume of space. But how could something have a physical presence, yet not respond to human touch or sight? Daniel Bernoulli, a Swiss mathematician, proposed an answer. If air – this substance that we could not see, feel, or smell – had weight, it must be made of something physical. In 1643, Evangelista Torricelli, an Italian mathematician and pupil of Galileo, showed that air had weight and was capable of pushing down on a column of liquid mercury (thus inventing the barometer). In the 17 th and 18 th centuries CE, several key events helped revive the theory that matter was made of small, indivisible particles. Because of Aristotle's great influence, Democritus's theory would have to wait almost 2,000 years before being rediscovered. Aristotle accepted the theory of Empedocles, adding his own (incorrect) idea that the four core elements could be transformed into one another. Ultimately, though, Aristotle and Plato, two of the best-known philosophers of Ancient Greece, rejected the theories of Democritus. This was a remarkable theory that attempted to explain the whole physical world in terms of a small number of ideas. Democritus theorized that atomos were specific to the material that they made up, meaning that the atomos of stone were unique to stone and different from the atomos of other materials, such as fur. He suggested that atomos were eternal and could not be destroyed. Democritus called these infinitesimally small pieces of matter atomos, meaning 'indivisible'. He reasoned that if you continued to cut the stone into smaller and smaller pieces, at some point you would reach a piece so tiny that it could no longer be divided. 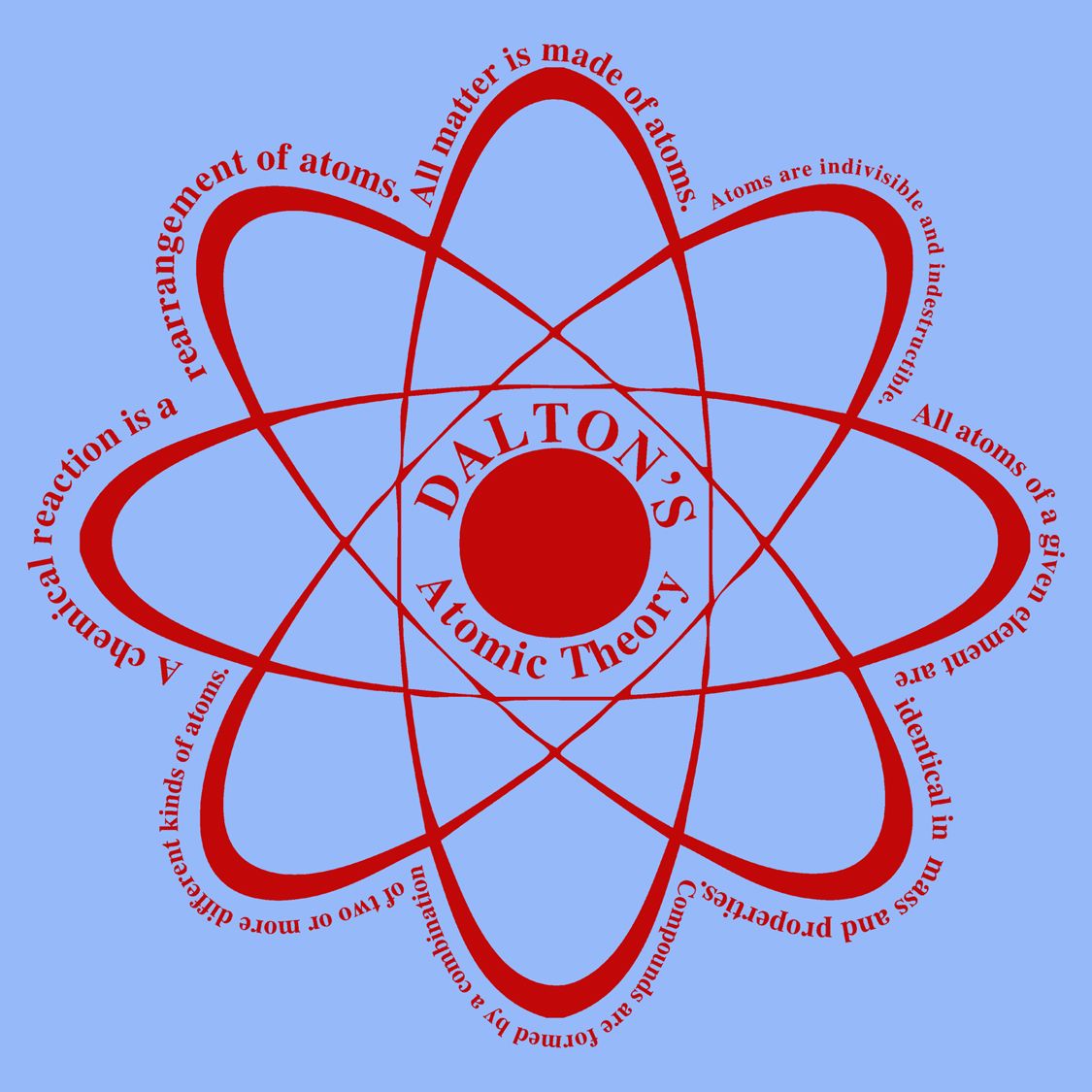
Democritus knew that if you took a stone and cut it in half, each half had the same properties as the original stone. 
Democritus's ideas were based on reasoning rather than science, and drew on the teachings of two Greek philosophers who came before him: Leucippus and Anaxagoras. Despite these problems, Empedocles's theory was an important development in scientific thinking because it was among the first to suggest that some substances that looked like pure materials, like stone, were actually made up of a combination of different "elements."Ī few decades after Empedocles, Democritus (460 BCE - 370 BCE), who was also Greek, developed a new theory of matter that attempted to overcome the problems of his predecessor. For example, regardless of how many times you break a stone in half, the pieces never resemble any of the core elements of fire, air, water, or earth. Empedocles's theory was quite popular, but it had a number of problems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
